|
7/23/2023 0 Comments Lead acid battery cathode reaction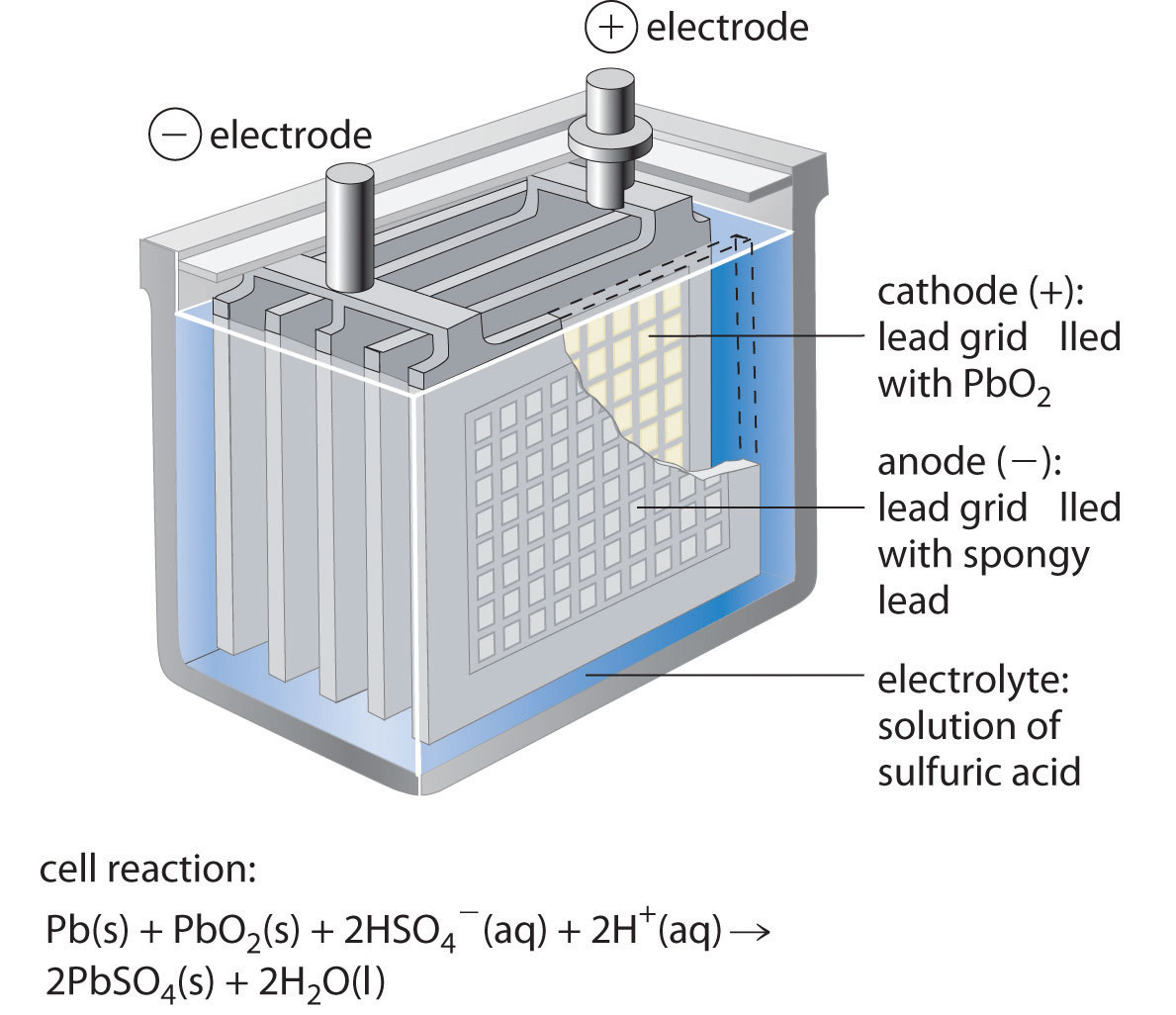 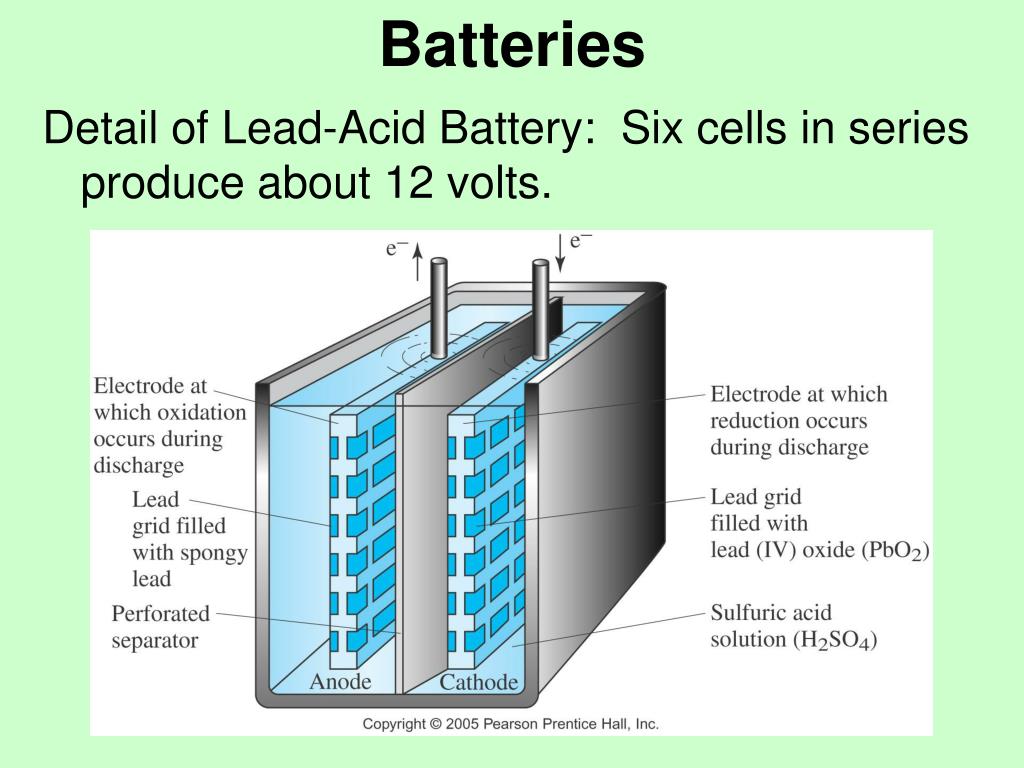 
The battery stores chemical energy in the form of a potential difference that exists between two poles of the battery immersed in an electrically conducting liquid solution. Larger turbine aircraft are usually powered by Nickel Cadmium (Ni-Cad) batteries which are more suited to starting turbine engines. A 12 V battery therefore contains 6 cells, while 24 V requires 12 cells.įigure 1: A battery installed on the firewall of a Cessna 152. The lead-acid batteries that form a part of these systems are built up from cells each outputting 2 V and connected in series to provide the desired voltage. Most light aircraft operate on either a 12 V or 24 V electrical system. Lead-acid batteries are heavy (they have a low energy-to-weight ratio) and large (low energy-to-volume ratio), but their ability to deliver a high current for a short period of time makes them ideally suited to power an internal combustion starter motor, which requires a short burst of high current to start the engine. Lead-acid starter batteries make up approximately 20 % of all battery sales second only to lithium-ion batteries found in cell-phones and laptops. Lead-acid batteries first appeared in the nineteenth century, yet they remain one of the most prevalent battery technologies in use today: primarily as a starter battery for internal combustion engines. Now we turn out attention to the battery – specifically the lead-acid battery which is the most commonly installed battery among general aviation aircraft. In the previous post we discussed the alternator which supplies electricity to the aircraft during engine operation. This is the third and final post in a three-part mini-series on the aircraft electrical system.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
